TISSEEL Fibrin Sealant

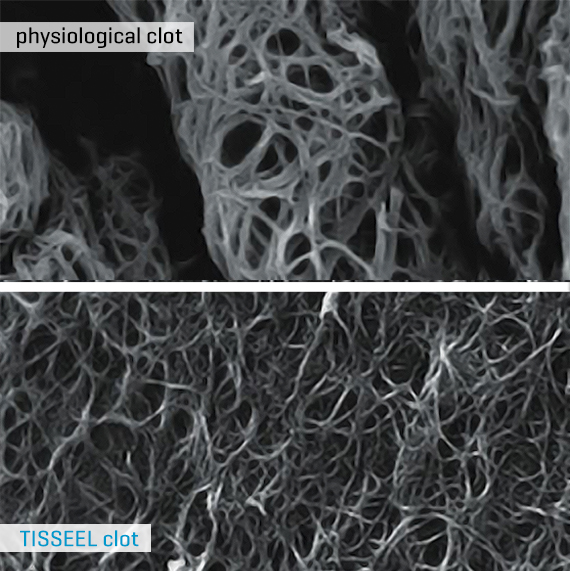
Mimics Physiological Clot
TISSEEL contains a unique formulation of fibrinogen and thrombin which, upon mixing, mimics a physiological clot1. The aprotinin in TISSEEL increases resistance of the fibrin sealant clot to degradation in a fibrinolytic environment1.
Mechanism of Action
Upon mixing, human sealer protein and human thrombin combine into fibrin that adheres to the wound surface and achieves haemostasis and sealing. TISSEEL mimics the final coagulation cascade step by forming a clot1. Learn more about how TISSEEL works.
Additional Product Benefits
Wide Range of Applicators
TISSEEL is the only fibrin sealant to offer a complete portfolio of drip and spray applicators, allowing surgeons to select the right tools to address individual patient needs1.
Precise Placement
The DUPLOSPRAY System allows for bleeding to be targeted laparoscopically by getting as close as 2-5 cm to the tissue¹.
Do not spray TISSEEL where the recommended minimum distance from the applicator tip to the target site cannot be assured. Consult your country-specific SPC or package leaflet for recommended distances and pressure.
Prescribing Information:
| Ireland TISSEEL |
Suspected Adverse Reactions and any drug product quality complaints (including suspected defective medicines) should be reported to the Health Products Regulatory Authority (HPRA) using a Yellow Card obtained from the HPRA, via the online system (www.hpra.ie) or by telephone on 01-6764971.
Adverse Events relating to Baxter products can also be reported direct to Baxter Pharmacovigilance on +44 1635 206360, or by email to [email protected]
Any drug product quality complaints relating to Baxter products can be reported directly to the Baxter Country Quality Assurance Team on 01 2065500, or by email to [email protected]. Alternatively please report directly to your Baxter Representative, who will take the details and forward to the Baxter Country Quality Assurance Team.